Results show a risk factor for AHPND in Pacific white shrimpResults of this study demonstrated that shrimp initially\r\nexposed to WSSV allow VpAHPND to cause faster and higher mortality than in\r\nsh...

Results show a risk factor for AHPND in Pacific white shrimp
Results of this study demonstrated that shrimp initially\r\nexposed to WSSV allow VpAHPND to cause faster and higher mortality than in\r\nshrimp infected with VpAHPND alone. Photo by Fernando Huerta.
Acute hepatopancreatic necrosis disease (AHPND), caused by\r\nspecific strains of the bacterium Vibrio parahaemolyticus (VpAHPND),\r\nhas been reported in shrimp cultured in Southeast Asia and the Americas since\r\n2009. The clinical signs of the disease include atrophied hepatopancreas (HP),\r\nan empty gastrointestinal tract, a milky appearance of the stomach and\r\nlethargy. This bacterial disease has caused up to 100 percent mortality in\r\npopulations of farmed Pacific white shrimp (Penaeus vannamei) and black tiger\r\nshrimp (P. monodon), and collective economic losses due to AHPND and other\r\nshrimp diseases are estimated at $23.6 billion globally with a further loss of\r\n$7 billion in feed sales.
In some cases, VpAHPND has been reported in\r\nco-infection with the spore-forming microsporidian Enterocytozoon\r\nhepatopenaei (EHP). Although EHP does not appear to cause mortality,\r\nEHP-infected animals would be more vulnerable to AHPND, which may cause\r\nsubstantial mortality in shrimp ponds. And EHP-infected shrimp have an\r\nincreased susceptibility to VpAHPND, suggesting that shrimp weakened by EHP\r\nwould succumb to secondary pathogens, such as VpAHPND.
For several years, white spot syndrome virus (WSSV) has\r\nspread worldwide throughout much of Asia, the Americas, Europe, Africa, Middle\r\nEast and Australia. In some cases, WSSV has been detected in healthy animals\r\nwithout clinical symptoms or mortalities, indicating that infection by WSSV\r\ndoes not necessarily result in high mortality, and secondary infection by\r\nopportunistic pathogens, such as Vibrio spp., would expedite the\r\nonset of the disease.
In 2017, P. vannamei mortality rates of up to 60\r\npercent were reported in the Philippines. The shrimp exhibited clinical signs\r\nof AHPND but were confirmed by PCR to be positive for both VpAHPND and\r\nWSSV. From this case, we hypothesized that WSSV infection without clinical\r\nsymptoms would allow VpAHPND to cause faster and higher mortality of pond\r\ncultured shrimp than those infected with VpAHPND alone.
This article – adapted and summarized from the\r\noriginal – reports on a study designed to support this hypothesis by\r\nsimulating the co-infection with WSSV and VpAHPND in juvenile shrimp P.\r\nvannamei under laboratory conditions. As a result, we demonstrated that\r\nshrimp initially exposed to WSSV were easily infected by VpAHPND, but hardly\r\nrecovered from the disease according to histopathology examination. In\r\naddition, using qPCR assay and immunohistochemistry [IHC; the most common\r\napplication of immunostaining, involving the process of selectively identifying\r\nantigens (proteins) in cells of a tissue section using the binding of\r\nantibodies specifically to antigens in biological tissues] examination, we\r\ndetermined that WSSV infection could be accelerated by the secondary VpAHPND infection.\r\nThis work was supported by the National Research Foundation of Korea (NRF)\r\ngrant funded by the Korea government (MSIT) (grant number\r\nNRF-2018R1C1B5086350).
Study setup
Postlarval P. vannamei were sourced from a local\r\nshrimp farm (Jeju Province, South Korea) and transported to the Institute of\r\nMarine Sciences of Jeju National University in South Korea. Shrimp were reared\r\nto an average weight of 0.5 ± 0.05 grams in 96-liter maintenance tanks with\r\naerated artificial seawater at 25 to 28 degrees-C and 30 ppt salinity. For the\r\nco-infection experiment, juvenile shrimp (average 0.5 grams, N=80) were\r\nrandomly selected from the maintenance tanks and distributed into experimental\r\ntanks. Before the co-infection experiment, representative shrimp were randomly\r\nselected and confirmed to be negative for both WSSV and VpAHPND by PCR.
For WSSV stocks, moribund P. vannamei with white\r\nspots on their cuticles were collected from an anonymous shrimp farm in\r\nChungcheong Province located on the western coast of Korea. For VpAHPND bacterial\r\nstocks, the strain 13–028/A3 was previously isolated from the stomach of\r\nmoribund P. vannamei from a farm in Vietnam and displaying whitish\r\nhepatopancreases (HP).
For detailed information on the experimental designs for\r\nWSSV and VpAHPND co-infection; qPCR assays; IHC examination of the gill\r\ntissues; histopathology examination of the HP tissues; and statistical\r\nanalyses, please refer to the original publication.
Results and\r\ndiscussion
We studied co-infection with WSSV and VpAHPND in\r\njuvenile P. vannamei shrimp under laboratory condition, and the\r\nmortality rates were compared with single infection groups.
Our results showed that co-infection accelerated shrimp\r\nmortality compared with single infection (Fig. 1). We observed 0 percent\r\nmortality Group 1 (WSSV only), and cumulative mortality was 50 percent at day 4\r\npost-infection (p.i.) in Group 2 (VpAHPND only). However, the mortality\r\nrate increased significantly in Group 3 (WSSV+VpAHPND) compared with the other\r\ngroups (p < .05). In Group 3, the shrimp began to show mortality at day 2\r\np.i., and cumulative mortality reached 75 percent at day 3 p.i. During the\r\n10-day experimental period, no mortality was observed in the negative control\r\n(Group 4, not challenged).
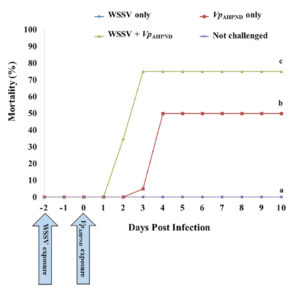
Fig. 1: Cumulative mortality of P. vannamei after\r\nchallenge with WSSV and VpAHPND (Group 1: WSSV only; Group 2: VpAHPND only;\r\nGroup 3: WSSV + VpAHPND; Group 4: not challenged). The mortality rate was\r\nsignificantly increased in the co-infection group (Group 3) compared with the\r\nsingle infection groups (Groups 1 and 2). Different letters indicate\r\nsignificant differences in survival at the termination day (day 10) (P\r\n< .05).
Our results also showed that co-infection increased the\r\nlevel of WSSV infection, which was confirmed via qPCR and IHC, and the levels\r\nof WSSV infection were markedly different between Group 1 (WSSV only) and Group\r\n3 (WSSV + VpAHPND) on the termination day (Day 10). Based on qPCR analysis,\r\nWSSV was detected in shrimp in Group 3 (WSSV+VpAHPND) but WSSV was negative in\r\nshrimp exposed to WSSV only (Group 1). Also, by IHC examination, brown\r\ncoloration against pink cytoplasm and purple nuclei, indicative WSSV-positive\r\nreactions were observed in the shrimp of Group 3 (WSSV+VpAHPND) (Fig. 2D)\r\nsimilar to the WSSV-positive sample previously prepared in the preliminary\r\nstudy (Fig. 2B). However, these WSSV-positive reactions were not seen in the\r\nshrimp exposed to WSSV alone (Group 1; Fig. 2A) and the negative control (Group\r\n4, not challenged; Fig. 2C).
Co-infection decreased the recovery of VpAHPND infection.\r\nAll experimental shrimp showed typical clinical symptoms of AHPND, including\r\npale HP and empty gut, within three to four days after infection in Group 2 (VpAHPND only)\r\nand Group 3 (WSSV+VpAHPND), and VpAHPND infection was confirmed by qPCR\r\nand histopathology examination. From the qPCR analysis, VpAHPND was\r\ndetected in dead shrimp collected from both Group 2 (VpAHPND only) and\r\nGroup 3 (WSSV+VpAHPND) on Day 3 and 4.
By histopathology examination, representative shrimp from\r\nboth Group 2 (VpAHPND only) and Group 3 (WSSV + VpAHPND) showed a typical\r\nterminal phase, characterized by massive bacterial infection and sloughing of\r\nhepatopancreatic tubule epithelial cells at the level of G3-4 (grade 4 is the\r\nmost severe) on Day 3.
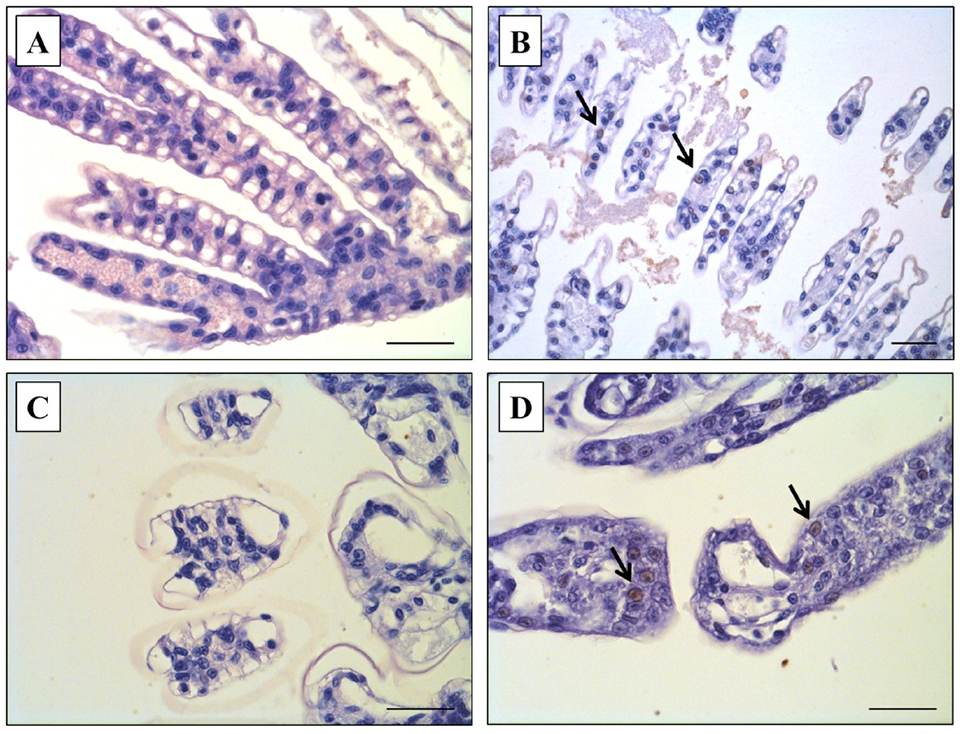
Fig. 2: Immunohistochemistry (IHC) examination from shrimp\r\ninfected with white spot syndrome virus (WSSV). (A) Negative control (Group 4):\r\nno WSSV-infected cells in gills of P. vannamei. (B) Positive control:\r\nWSSV-positive cells in gills of P. vannamei infected with WSSV tissue\r\nby the feeding method. (C) WSSV control (Group 1): no WSSV-infected cells in\r\ngills of P. vannamei infected with WSSV by the immersion method. (D) WSSV and\r\nVpAHPNDco-infection (Group 3): WSSV-positive cells in gills of P. vannamei pre-infected\r\nwith the WSSV and post-challenged with the secondary pathogen, VpAHPND.\r\nHematoxylin counterstain. Scale bar: 40 μm.
However, on the termination day (day 10), a remarkable\r\ndifference in the histopathology of the HP of surviving shrimp was seen between\r\ngroup 2 (VpAHPND only) and Group 3 (WSSV+VpAHPND). Histopathology signs of\r\nthe group 2 (VpAHPND only) survivors had an HP of normal appearance, and\r\nnormal functions of B and R cells (Fig. 3B), the same as the negative control\r\n(Group 4, Fig. 3A). To the contrary, Group 3 (WSSV+VpAHPND) survivors showed\r\ntypical AHPND histopathology signs in the HP, with massive bacterial infection\r\nand sloughing of hepatopancreatic tubule epithelial cells of G4 severity (Fig.\r\n3C).
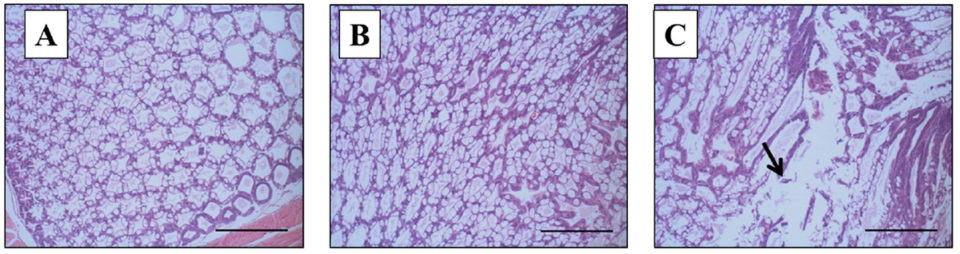
Fig. 3: Histopathology examination using Davidson’s\r\nAFA-fixed tissue sections from shrimp infected with VpAHPND, on the termination\r\nday (Day 10). (A) Negative control (Group 4): normal appearance of the\r\nhepatopancreas. (B) AHPND control (Group 2): normal appearance of the\r\nhepatopancreas with normal function of B and R cells. (C) WSSV and VpAHPND\r\nco-infection (Group 3): the typical acute phase with progressive necrosis and\r\nmassive sloughing of hepatopancreatic tubule epithelial cells. Mayer-Bennett’s\r\nhematoxylin/eosin-phloxine (H&E) staining. Scale bar: 180 μm.
Infections with multiple pathogens (co-infection) are common\r\nin practical shrimp cultures. Several cases of shrimp vibriosis with WSSV\r\ninfection have been reported with various penaeid species in different\r\ncountries. There has been no prior detailed analysis on the roles of these\r\npathogens in shrimp co-infected with WSSV and Vibrio. In shrimp farms, the\r\nWSSV infection without symptoms or mortality probably causes shrimp weakening\r\nand might trigger the VpAHPND infection.
We also observed declines in antioxidant and\r\nantiperoxidative enzymes in shrimp infected with VpAHPND, similar to WSSV in\r\nour ongoing study, and we suspect that VpAHPND infection may also have\r\ncompromised the animals, increasing their susceptibility to other pathogens in\r\nponds.
Intramuscular (IM) routes have been widely used for\r\nexperimental infection of shrimp with WSSV. However, IM injection has not been\r\nconsidered as an infection route of VpAHPND. In addition, per os feeding can\r\nproduce both WSSV and VpAHPND infection, but this is not a practical\r\nmethod for the dual-infection experiments because experimental shrimp were\r\nsupposed to be infected twice within short intervals. Therefore, for the\r\ncurrent bioassay, we selected the immersion (waterborne) method to co-infect\r\nshrimp with WSSV and VpAHPND. The synergistic effects influenced by the same\r\ninfection routes have already been confirmed between WSSV and Vibrio spp.
Perspectives
To the best of our knowledge, our study is the first to\r\nexamine the co-infection of WSSV and VpAHPND under laboratory conditions.\r\nIn ponds, shrimp that were infected with WSSV (or other pathogens) did not\r\nalways show clinical signs or mortalities. In those cases, even WSSV-positive\r\nshrimp without any clinical symptoms may cause higher and faster mortality than\r\ntheir disease-free counterparts, and the shrimp would hardly recover from the\r\ndisease following a secondary infection, such as VpAHPND.
\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n
Also, the secondary infection would provoke WSSV infection\r\nor virus replication, as confirmed in the present study. Therefore, shrimp\r\nfarmers should pay attention to the management of multiple infections (WSSV or\r\nother pathogens) in their ponds to prevent further losses in shrimp production.
Source : Global Aquaculture Alliance

Ditulis oleh
Tim Minapoli
Kontributor
Pakar di bidang akuakultur dengan pengalaman lebih dari 15 tahun. Aktif berkontribusi dalam pengembangan industri perikanan Indonesia.
