A recent study by Nguyen Dinh Thang et al. 2019, for the\r\nfirst time, found that food supplemented with melanin helps protect whiteleg\r\nshrimp from the white spot syndrome virus (WSSV). The r...
A recent study by Nguyen Dinh Thang et al. 2019, for the\r\nfirst time, found that food supplemented with melanin helps protect whiteleg\r\nshrimp from the white spot syndrome virus (WSSV). The report is published\r\nin International Aquatic Research.
Whiteleg shrimp ( Litopenaeus vannamei )\r\nis a major species in South Africa and Asia, especially Brazil, Ecuador,\r\nMexico, China, Thailand, Indonesia and Vietnam. However, viral diseases\r\nare a serious problem for shrimp industry in these countries. White spot\r\nsyndrome virus (WSSV) has been considered as one of the most dangerous\r\npathogens that infect shrimp, causing mortality as high as 100% within 7-10\r\nvirus infections.
Shrimp immunity is highly dependent on the innate immune system\r\nincluding cellular barrier and humoural these barriers play an important role\r\nin protecting shrimp from pathogen attack. In addition, melanization is\r\nalso an important immune mechanism in shrimp.
Melanin is a naturally occurring polymer with very low\r\ntoxicity (Garcia Borron et al. 2014). Melanin has many important roles in\r\nthe human body including absorption of UV radiation, antioxidants and heavy\r\nmetal absorption. Melanization and melanin secretion help shrimp protect\r\nthemselves by packaging pathogens and excreting them into the\r\nenvironment. However, no studies have examined the role of melanin as a\r\nsupplement in protecting shrimp from pathogen attacks. Therefore, in this\r\nstudy, it was the first time to create feed containing melanin and apply them\r\nto vannamei ( Litopenaeus vannamei ) farmed to investigate\r\nits ability to protect shrimp against WSSV.
Feed containing\r\nmelanin protects vannamei from WSSV virus
Prepare the food
Natural Melanin is extracted from Squid (Loligo\r\nformosana). Commercial granular feed with a diameter of 2 mm is used as\r\nthe main feed to produce feed containing melanin with different\r\nformulations.
Formula 1 (F1): Melanin is coated on the surface of\r\ncommercial feed particles using cod oil as a binding agent. At first,\r\nmelanin powder (size <150m) was distributed into cod oil and evenly\r\ndistributed on commercial feed particles at different ratios (w / w) 1/500,\r\n1/200 and 1 / 50.
Formula 2 (F2): Melanin is mixed with commercial\r\nflour and starch solution (3%) used as a binder. First, commercial feed\r\nparticles are ground into a powder. After that, commercial food flour and\r\nmelanin powder are mixed at different ratios of 1/500, 1/200 and 1/50 (w / w)\r\nand evenly distributed in 3% starch solution. Next, the mixture is\r\nextruded to form particles with a diameter of 2 mm.
Formula 3 (F3): Commercial feed particles are dipped\r\ndirectly into the ink liquid at 1/1 (w / v).
Although F1, F2 and F3 feeds were originally created with\r\ndifferent levels of melanin / feed , including 1/500, 1/200 and 1/50,\r\nthe preliminary results of the researchers show. that the 1/200 ratio is\r\nthe best fit and therefore researchers used the feed at this ratio for\r\nsubsequent experiments.

A) The dose of WSSV causes 75% of shrimp to\r\ndie. Mortality was challenged with different doses of WSSV; B)\r\nMorphology of negative control (NC) and WSSV-infected shrimp (PC) on day 1\r\n(D1), day 4 (D4), day 7 (D7), day 10 (D10) and day 14 (D14) ; C)\r\nExpression of the VP28 gene in PC and NC shrimp after viral infection. The\r\nblue and red circles indicate necrotic heads and red tails occurring on\r\nWSSV-infected shrimp.
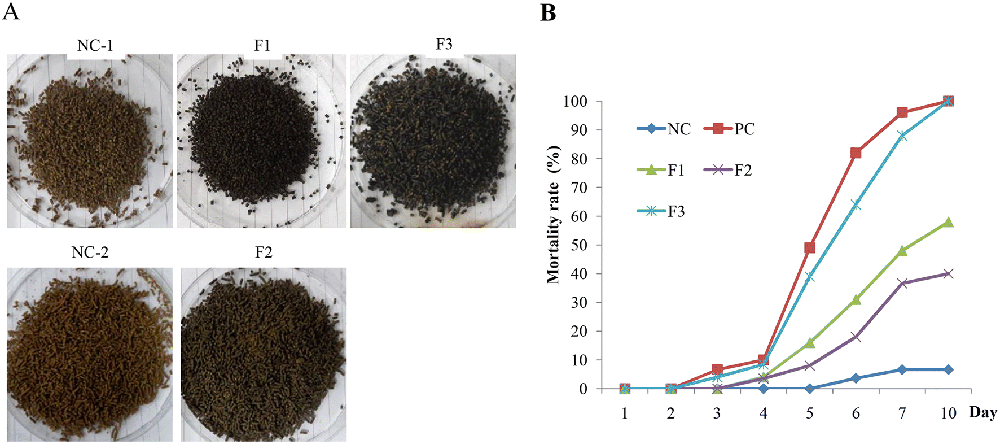
Shrimp were raised and fed with F1, F2 or F3 feed, then\r\nchallenged with WSSV for 3 days. Dead shrimp are collected and counted.
The results demonstrate that feed preparation formula\r\ncontaining melanin affects the protective ability of melanin\r\nsupplementation in cultured shrimp . Compound feed F2 has a\r\nsignificantly higher protection rate than feed coated with melanin (F1 and\r\nF3). Mixing F2 form can obtain melanin inside feed particles, while\r\nformulas coated with F1 and F3 cannot keep melanin in water; Therefore,\r\nthe amount of melanin consumed when raising F2 shrimp is much higher than that\r\nof F1 or F3 cultured shrimp. This result can lead to differences in the\r\nprotection capacity of feed types. The results of this study showed that\r\nthe protection rate depends on the amount of melanin that shrimp have consumed; therefore,\r\nproviding F2 and F3 feed mixtures for shrimp does not appear to have a\r\nsynergistic effect on the rate of protection.

The amount of melanin consumed by shrimp depends on the type\r\nof feed. WSSV-infected shrimp were fed with NC1, F1 and F2 feed,\r\nrespectively. The red circles indicate different amounts of melanin in the\r\nshrimp gut. D) Melanin is released from food into the environment.
The results of this study indicated that protection rates\r\nfor shrimp fed F2 feed were about 60, 65%. This result is very impressive,\r\nbut reasonable; Melanin supplementation can only partially help shrimp\r\ncatch and pack the invasive virus through the digestive system, but it cannot\r\nhelp eliminate the virus that causes shrimp disease by other means of\r\npenetration. Therefore, the simultaneous use of other substances that can\r\nstimulate the activity of the immune system may be necessary. Moreover,\r\nthis study used natural melanin extracted from ink bags, which is considered a\r\nwaste from seafood processing plants. Therefore, using this waste source\r\nto produce melanin will bring great benefits because of its low cost.
\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n
The results showed that F2 had a 64% protection rate on\r\nSaturday and 62% on day 10 after the virus challenge. The protective\r\nability of feed depends on the amount of melanin consumed by\r\nshrimp. Moreover, Gen VP28, VP28 (28 kDa) is an important structural\r\nprotein of WSSV that can be used as a marker to determine the presence of WSSV\r\nin water or / and shrimp samples, which have provided presence of WSSV, was\r\nsignificantly reduced in F2 shrimp.
Source : tepbac.com

Ditulis oleh
Tim Minapoli
Kontributor
Pakar di bidang akuakultur dengan pengalaman lebih dari 15 tahun. Aktif berkontribusi dalam pengembangan industri perikanan Indonesia.
