In an aquaculture system, shrimp and microorganisms share\r\nthe same aquatic medium; thus, the intestinal microbial community interacts\r\ndirectly with planktonic microbiota. Therefore, characteriza...
In an aquaculture system, shrimp and microorganisms share\r\nthe same aquatic medium; thus, the intestinal microbial community interacts\r\ndirectly with planktonic microbiota. Therefore, characterization of the\r\nintestinal microbiota (IM) of aquatic organisms is a priority to understand\r\nhost-microorganism interactions and the corresponding relationship with the\r\nsurrounding microbiota.
Numerous strategies have been developed to improve the\r\ncolonization of the gastrointestinal tract of aquatic animals with beneficial\r\nbacteria and avoid proliferation of pathogenic bacteria. One of these\r\napproaches has been diets with pre‐, pro‐ and symbiotic supplementation, which\r\ncan also improve animal growth and feed efficiency. Probiotics have shown to be\r\na promising and environmentally friendly alternative for disease prevention,\r\nespecially in the culture of high-value crustaceans like shrimp.
Several studies have indicated that probiotics can\r\ncontribute to enzymatic digestion, inhibit pathogenic microorganisms, promote\r\ngrowth factors and increase the immune response of aquatic organisms.\r\nConsequently, new beneficial microorganisms that could be used as probiotics in\r\naquaculture are constantly explored. Marine actinomycetes are among those\r\npromising candidates by virtue of their ability to produce a wide variety of\r\nantibiotics and extracellular enzymes.
This article – adapted and summarized from the original (J.M.\r\nMazón-Suástegui et al. 2019. Effect of Streptomyces probiotics on the\r\ngut microbiota of Litopenaeus vannamei challenged with Vibrio\r\nparahaemolyticus. Microbiology Open Vol. 9, Issue 2) – reports on a study to\r\ndetermine the effect of Streptomyces strains on the intestinal\r\nbacterial community in juveniles of the Pacific white shrimp, as part of a\r\nprevious study revealing the probiotic effect of Streptomyces strains\r\nalone or combined with Bacillus and Lactobacillus.
This study was financed by Sectoral Fund for Research and\r\nEducation of México: Projects CONACyT Basic Science 258282 and Proinnova\r\nCONACyT 241777, under the academic responsibility of JMM‐S. We thank the\r\nfollowing CIBNOR staff: Patricia Hinojosa, Delfino Barajas and Pablo Ormart,\r\nand Diana Fischer for their editorial services.
Study setup
Four experimental groups of shrimp with three replicates\r\neach were used in the experiment, treated with probiotic agents as follows: (a)\r\nRL8 (Streptomyces sp. RL8); (b) Lac‐Strep (Lactobacillus graminis + Streptomyces sp.\r\nRL8 and Streptomyces sp. N7; 1:1:1 proportion); (c) Bac‐Strep (B.\r\ntequilensis YC5‐2, B. endophyticus C2‐2, B. endophyticus YC3‐B, Streptomyces sp.\r\nRL8, and Streptomyces sp. N7; 1:1:1:1:1 proportion); and (d) control\r\ngroup (no probiotics added).
The experimental shrimp were fed with a commercial, 35\r\npercent protein, pelletized feed (Purina®, Ciudad Obregón, Sonora, Mexico) in\r\nwhich the probiotic suspensions were incorporated by spraying. The Lactobacillus and Bacillus strains\r\nwere incorporated at a final concentration of 1 × 106 colony‐forming units\r\n(CFU) per gram of feed, whereas Streptomyces strains were added at a\r\nratio of 1 × 108 CFU per gram of feed, which is the mean of the dose range\r\nused for most of the probiotics.
Treated shrimp were fed ad libitum three times a\r\nday over the course of 30 days with the probiotic‐sprayed commercial diets, and\r\nthe control group was fed with the commercial diet sprayed with sterile\r\nseawater. The bacterial load in the feed was confirmed by plate count;\r\nparticulate matter was removed daily by siphoning during the probiotic feeding\r\nperiod followed by the addition of discarded water (25 percent). No water\r\nexchange was performed during the challenge, and dead animals were regularly\r\nremoved from tanks throughout the daylight hours.
For detailed information on the experimental design; test\r\norganisms; DNA extraction and sequencing; and statistical analyses, refer to\r\nthe original publication.
Results
In general, microbiota diversity in most of the experimental\r\ngroups was higher after than before the challenge with V. parahaemolyticus.\r\nFig. 1 shows the Principal Component Analysis (PCA, a tool for making\r\npredictive models and often used to visualize genetic distance and relatedness\r\nbetween populations) of the beta diversity (an ecological term for the ratio\r\nbetween regional and local species diversity) associated with microbiota\r\nvariance for groups treated with probiotics and control groups, before and\r\nafter V. parahaemolyticus challenge.
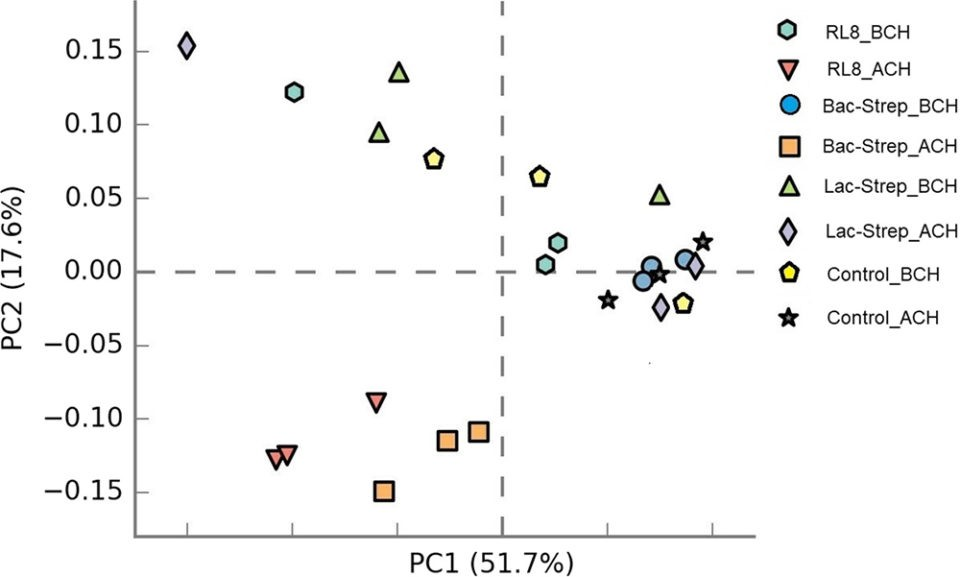
Fig. 1: Principal components analysis (PCA) plot of beta\r\ndiversity associated with the gut microbiota of L. vannamei treated\r\nduring 30 days with Streptomyces alone [RL8] and combined with Bacillus\r\n[Bac‐Strep] and Lactobacillus [Lac‐Strep]; before [ˍBCH] and after [ˍACH]\r\nchallenge with Vibrio parahaemolyticus. Adapted from the original.Results\r\nof our study showed that, after the challenge, the microbiota composition of\r\nshrimp treated with RL8 and Bac‐Strep was different from the rest of the\r\nexperimental groups, based on PCA. These treatments were grouped on the left\r\nside of the chart along the first principal component axis (PC1). In contrast,\r\nthe intestinal microbiota (IM) of shrimp from the rest of the groups showed a\r\nwider dispersion, indicating differences in beta diversity.
The composition and abundance of the bacterial community of\r\nthe different experimental groups is shown in Fig. 2. A total of 14 phyla were\r\nidentified in the intestine of L. vannamei: Proteobacteria,\r\nActinobacteria, Bacteroidetes, Verrucomicrobia, Firmicutes, Planctomycetes,\r\nFibrobacteres, Cyanobacteria, TM7, Chlamydiae, TM6, Chlorobi,\r\nFusobacteria, and GNO2. Except for Bac‐Strep_ACH, Proteobacteria was\r\nthe most abundant bacterial phylum in all experimental groups before and after\r\nchallenge, with an average relative abundance of 45.34 ± 6.0 percent and 58.62\r\n± 2.74 percent, respectively. This phylum was followed by Actinobacteria and Bacteroidetes with\r\nrelative abundances of 30.40 ± 3.11 percent and 21.21 ± 3.70 percent and, 22.15\r\n± 5.66 percent and 18.44 ± 0.73 percent before and after challenge,\r\nrespectively.
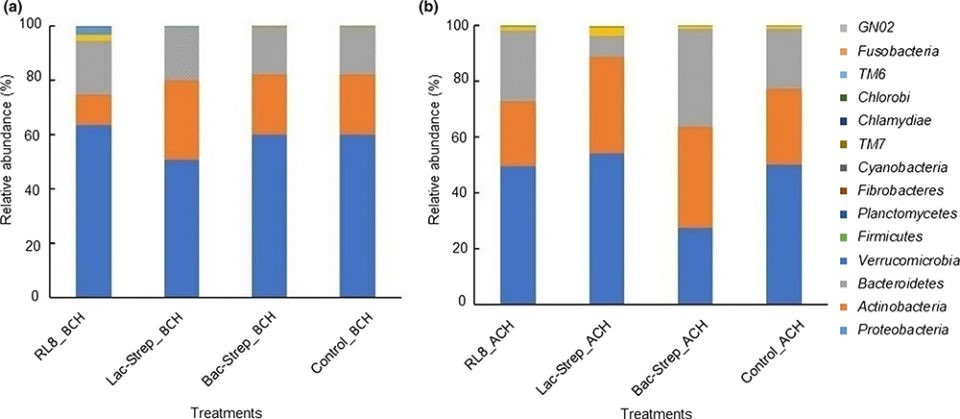
Fig. 2: Relative abundance (percentage of each phylum with\r\nrespect to all valid sequences for each treatment) of different bacterial phyla\r\nassociated with the gut microbiota of L. vannamei treated during 30\r\ndays with Streptomyces alone [RL8] and combined with Bacillus [Bac‐Strep] and\r\nLactobacillus [Lac‐Strep]; (a) before [ˍBCH] and (b) after [ˍACH] challenge\r\nwith V. parahaemolyticus.The three bacterial classes with the highest\r\nrelative abundance were Alphaproteobacteria with\r\n42.5 ± 5.82 percent; Actinobacteria with\r\n29.16 ± 3.30 percent; and Flavobacteriia with\r\n21.45 ± 5.65 percent before challenge (BCH), and\r\n35.23 ± 3.74 percent, 25.81 ± 6.35 percent, and\r\n18.37 ± 0.92 percent after challenge (ACH), respectively (Fig. 3).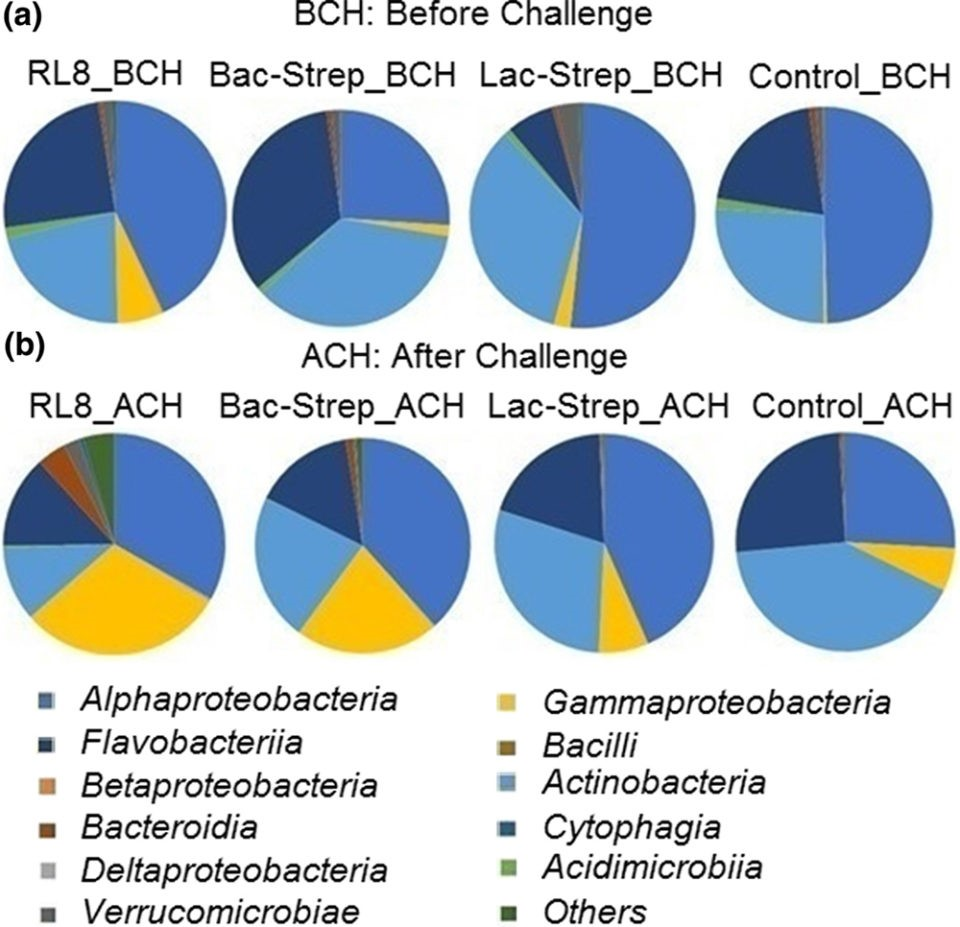
Fig. 3: Relative abundance (percentage of each class\r\ncompared with the total) at class level associated with the gut microbiota\r\nof Litopenaeus vannamei treated during 30 days with Streptomyces\r\nalone [RL8] and combined with Bacillus [Bac‐Strep] and Lactobacillus\r\n[Lac‐Strep]; (a) before [ˍBCH] and (b) after [ˍACH] challenge with V.\r\nparahaemolyticus.An animal’s intestine is a vital organ for food storage, nutrient\r\ndigestion and absorption in addition to playing an important role in immunity.\r\nSeveral intestinal functions are achieved through bacterial metabolism, which\r\nmay also benefit the host by improving the immune response, nutrient absorption\r\nand homeostasis maintenance. Consequently, modulation of the IM – through\r\noptimization of diet formulation or supplementation with pre- and probiotics –\r\nis important to improve the general physiological development and increase the\r\nproductivity and economic revenues during shrimp farming.
Results of this study showed that the groups RL8_ACH and Bac‐Strep_ACH\r\nhad higher bacterial diversity, which has been associated with a greater host\r\nresistance to pathogen colonization than the other experimental groups. Even\r\nthough a significant change in microbiota composition of L. vannamei has\r\nalso been achieved with other probiotics, this is the first study showing such\r\neffect with Streptomyces strains, either alone or combined. Results\r\nfor the Control_ACH group challenged with V. parahaemolyticus suggested\r\nless bacterial diversity and species richness due to the presence of the\r\npathogen and, thus, a greater susceptibility to invasion by this agent.
Proteobacteria was the dominant phylum in the intestine\r\nof L. vannamei treated with probiotics before and after V.\r\nparahaemolyticus challenge, followed by Actinobacteria and Bacteroidetes.\r\nThis phylum has been regarded as the most abundant in L. vannamei in\r\nmultiple studies with relative abundances from 68 percent to 97 percent.\r\nSimilar results were also reported at different salinities and food types.\r\nOther studies have detected the phyla Firmicutes, Bacteroidetes and Actinobacteria as\r\nthe most dominant after Proteobacteria. Nonetheless, the relative\r\nabundance of these bacteria in the intestine of L. vannamei changes\r\naccording to environment conditions and diet composition.
Actinobacteria was the second most abundant phylum in\r\nshrimp intestine. Some members of this phylum are known to be excellent\r\nsecondary metabolite producers that can protect the host from an infection.\r\nAdding Streptomyces strains to feed resulted in a greater survival\r\nof L. vannamei after V. parahaemolyticus challenge. Our\r\nresults confirm the great potential of Streptomyces strains as\r\nprobiotic agents in aquaculture.
Microbial colonization and survival in the intestines of\r\ntargeted organisms are usually claimed as crucial prerequisites for potential\r\nprobiotics. However, these conditions do not seem to be strictly required for\r\nshellfish organisms which can benefit from their continuous interaction with\r\nbeneficial microorganisms thriving in the water and sediment. This appears to\r\nbe the case for Streptomyces sp. RL8, which is indigenous to the sea\r\nsediment, grows at a wide range of pH and salt concentrations, and produces resistant\r\nspores along with several extracellular enzymes and antimicrobial metabolites.\r\nConsequently, the modulatory effect on shrimp microbiome found here is not\r\nsurprising, the same as several other probiotic effects already described for\r\nthis strain.
Perspectives
Results of our study revealed that Proteobacteria,\r\nActinobacteria and Bacteroidetes were the predominant phyla in the intestine\r\nof L. vannamei shrimp. It also showed the modulating effect of Streptomyces sp.\r\nRL8 on L. vannamei microbiota, as well as its stimulatory effect on\r\nseveral antimicrobial producers that protected shrimp from V.\r\nparahaemolyticus infection. These results contribute to a better\r\nunderstanding of the composition and dynamics of L. vannamei intestinal\r\nmicrobiota and to the development of novel probiotics for the culture of this\r\nshrimp species.
\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n
Source: Global Aquaculture Alliance

Ditulis oleh
Tim Minapoli
Kontributor
Pakar di bidang akuakultur dengan pengalaman lebih dari 15 tahun. Aktif berkontribusi dalam pengembangan industri perikanan Indonesia.
