Despite a number of challenges that still need to be\r\novercome, genome sequencing has a growing range of practical applications in\r\naquaculture and, as costs come down, it is going to be increasin...
Despite a number of challenges that still need to be\r\novercome, genome sequencing has a growing range of practical applications in\r\naquaculture and, as costs come down, it is going to be increasingly widely\r\nused.
It hardly seems like 20 years ago when the Human Genome\r\nProject announced that\r\nit had sequenced “a working draft” of the human genome. Genome sequencing has\r\nbecome faster and cheaper since the project began and we are now able to get\r\nour individual genomes sequenced by commercial labs. But what is genome\r\nsequencing and what, if anything, does it mean for aquaculture?
How genome sequencing was achieved for the Human Genome\r\nProject. Technologies vary, but the principle is the same for sequencing the\r\ngenomes of aquatic species too
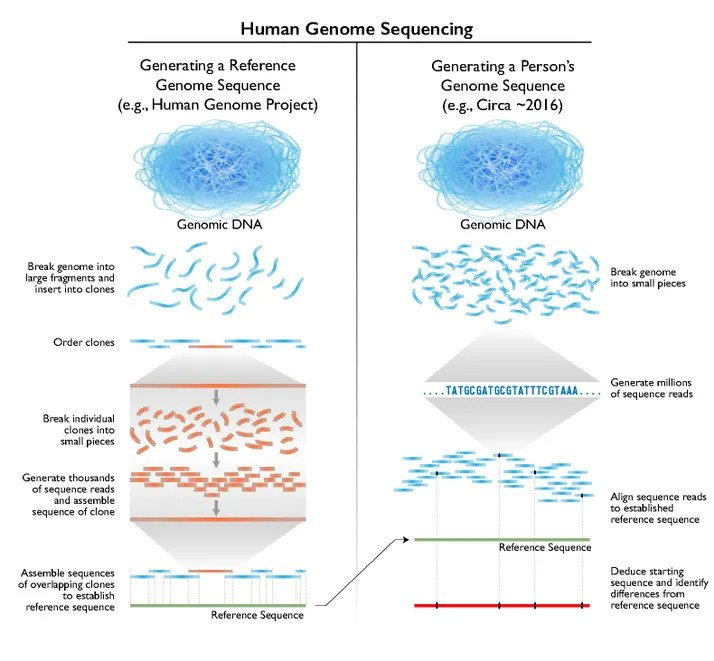
© Credit National Human Genome Research Institute
Background
The molecule DNA is often characterised as containing “the\r\nbuilding blocks of life”, with its long chemical sequences forming the genes\r\nthat control so many aspects of our physiology. Most of our genes are no\r\ndifferent from one person to the next, but within this uniformity there are\r\nsmall variations which give rise to some of the characteristics that make us unique,\r\nlike eye colour. The full suite of an individual’s genes is known as their\r\ngenome. Put simply, genome sequencing is a way for us to read the genes in a\r\ngenome.
Building up a library of genomes, we can find where there\r\nare variations between individuals, and unravel how these variations affect\r\ncharacteristics. We can also better understand how genes interact with each\r\nother, and even with the environment, to produce variations in characteristics\r\n– such as how temperature can influence the size of fish. We can also compare\r\ngenomes from one species to the next, seeing if the same genes control the same\r\ncharacteristics, or if similar variations or mutations lead to similar\r\noutcomes.
Although currently less prevalent in aquaculture than in\r\nterrestrial farming, embracing genetics is not new. Traditional selective\r\nbreeding methods rely on us looking at an individual’s phenotype – how its\r\ngenes are expressed in its environment. If we see an individual grows\r\nparticularly large, for example, we may want to use that individual for\r\nbreeding larger offspring. In fact, genetics underlies all sorts of\r\ncharacteristics of interest to aquaculturists.
Just over a year ago, Dr Hugues de Verdal, based at CIRAD in\r\nFrance, published his work demonstrating that feed\r\nefficiency is a heritable trait in Genetically Improved Farmed Tilapia\r\n(GIFT). In fact, de\r\nVerdal estimates that genetics explains around 30 percent of a tilapia’s\r\nfeeding efficiency, which is no small amount. Because the trait is heritable –\r\ndriven by the genes of the individuals – it is possible to selectively breed\r\nthe tilapia to promote feeding efficiency.

Dr Hugues de Verdal's research demonstrated that feed\r\nefficiency is a heritable trait in Genetically Improved Farmed Tilapia (GIFT)
© Hugues de Verdal
De Verdal’s work is among the first to identify the genetic\r\nunderpinnings of feed efficiency in aquatic species. Although he did not use\r\nsequencing in this study, de Verdal believes that embracing such techniques has\r\nsubstantial benefits, namely in terms of precision and predictive ability.
“You will have more information about the genes given by the\r\nparents, so it is more accurate to use genomic techniques to estimate the\r\nheritability [of a trait] and to estimate the breeding values of each fish,” he\r\nexplains.
Sometimes the genetic basis of characteristics is relatively\r\neasy to isolate. Take antifreeze protein genes, for example, which allow some\r\nspecies to survive in sub-zero temperatures. Experimental\r\nantifreeze-protein-gene transfer from winter flounder into fertilised Atlantic\r\nsalmon eggs was part of the early stages of transgenic salmon research.
Most traits that aquaculturists desire, however, do not come\r\nfrom a single gene. De Verdal suspects that feed efficiency arises from a suite\r\nof genes.
“We are quite sure there is no one gene which is leading all\r\naspects of feed efficiency because it could come from a lot of different\r\nthings, such as assimilation of nutrients, potentially the size of the\r\ngastrointestinal tract and expiration,” he explains. “It's probably really\r\ncomplex.”
Genome sequencing helps untangle such complexities. The\r\npotential applications for aquaculture are many. For the hatchery operator\r\naiming for a monosex population, understanding how genes and the environment\r\ninteract on a genotypic rather than phenotypic level can, for example,\r\nprecisely guide choice of temperature, which influences sex in some species.\r\nFor breeders, advanced selective breeding techniques allow for efficient and\r\nprecise development of strains specifically cultivated for a range of farming\r\nconditions. It may even be possible to manipulate genes that control\r\ncharacteristics like disease resiliency. Some researchers have even started\r\nsequencing pathogen genomes which, among other things, can help identify transmission\r\npathways, helping improve disease management. For those who use wild\r\nbroodstock, understanding the genome of the wild population can not only help\r\nmanage the broodstock fishery, but also ensure that you haven’t accidentally\r\npicked up a species that looks like the one you want, but actually is something\r\nelse.
This is just a small sample of the potential applications,\r\nthe number of which will invariably rise as aquatic-species genomic research\r\nbecomes more commonplace.

A machine used to sequence genomes
© Kenneth Rodrigues
Challenges
As with any technological application, there are challenges,\r\nsome of which are unique to aquatic species. As Professor Kyall Zenger at James Cook University in Australia\r\nhighlights, this includes the relatively high levels of polysaccharides found\r\nin animals such as crustaceans and shrimps.
“Polysaccharides are a complex compound used for defence\r\nmechanisms and other biological processes, but they inhibit sequencing,” Zenger\r\nexplains. “It’s not as simple as extracting [genomes] from a terrestrial animal\r\nthat has been routinely sequenced.” In fact, for most aquaculture species,\r\nthere is no baseline genome sequence from which researchers can begin working.\r\nBoth Zenger and de Verdal point out that building a baseline of any species can\r\nmean sequencing the genome of thousands of individuals.
A further challenge relates to the highly repetitive genomes\r\nof aquatic species – and their high rates of mutations. When a genome is\r\nsequenced, we do not get a single, neatly ordered output. Genomes are sequenced\r\nin small parts, which are then put back together in the correct order. “The\r\nmore complex the genome and the more repetitive it is, the more difficult it is\r\nto put back together because the [sequencing] software gets confused, so to\r\nspeak,” Zenger explains.
Zenger notes that genomic technology applications on aquatic\r\nspecies are still in the early stages of development and is confident that the\r\ntechnological challenges – and the lack of baseline sequences – will be\r\novercome.
For aquaculturists eager to start using genome sequencing\r\ndirectly in their farms or hatcheries, there is some bad news.
“You can’t buy the technology off the shelf,” Zenger points\r\nout. Furthermore, although the cost involved in sequencing is declining, making\r\nresearch applications more accessible, it currently remains too high for many\r\naquaculturists – except for large commercial operations working with high-value\r\nspecies.
\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n
This, however, may be a temporary situation. Current\r\nresearch continues to form the basis for commercial applications, and costs\r\ncontinue to decline. Routine commercial use may not be that far beyond the\r\nhorizon.
Source: The Fish Site

Ditulis oleh
Tim Minapoli
Kontributor
Pakar di bidang akuakultur dengan pengalaman lebih dari 15 tahun. Aktif berkontribusi dalam pengembangan industri perikanan Indonesia.
